Comments due March 28
Many large research universities have significant medical research and healthcare delivery enterprises. The leadership of those enterprises discount the effect of standards like this at their peril. It is easy to visualize that this document will have as transformative effect upon the healthcare industry as the ISO 9000 series of management standards in the globalization of manufacturing.
Standardization in the field of healthcare organization management comprising, terminology, nomenclature, recommendations and requirements for healthcare-specific management practices and metrics (e.g. patient-centered staffing, quality, facility-level infection control, pandemic management, hand hygiene) that comprise the non-clinical operations in healthcare entities.
Excluded are horizontal organizational standards within the scope of:
-
- quality management and quality assurance (TC 176);
- human resource management (TC 260);
- risk management (TC 262);
- facility management (TC 267), and;
- occupational health and safety management (TC 283).
Also excluded are standards relating to clinical equipment and practices, enclosing those within the scope of TC 198 Sterilization of health care products.
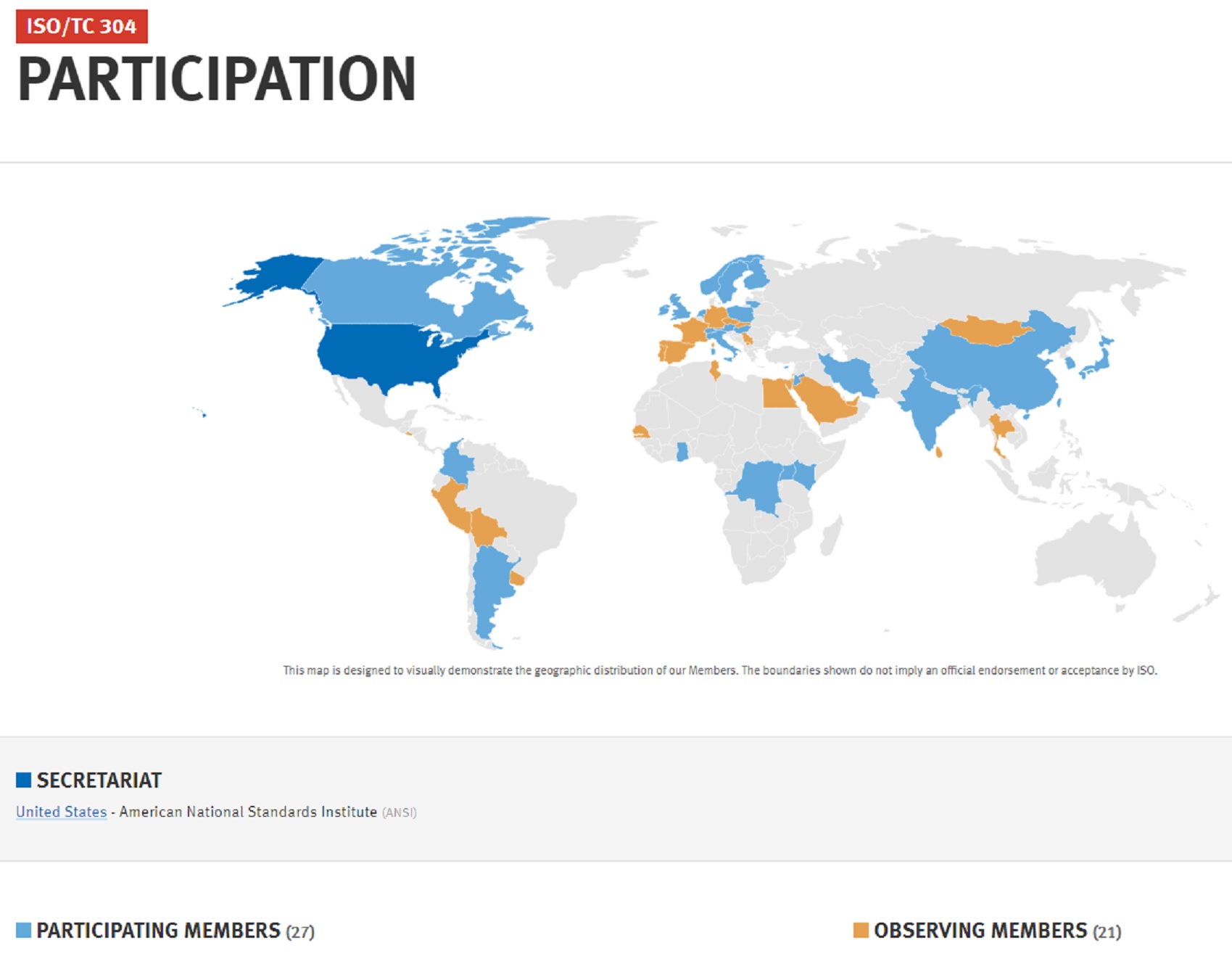
This committee is led by the US Technical Advisory Group Administrator —Ingenesis. The committee is very active at the moment, with new titles drafted, reviewed and published on a near-monthly basis,
DPAS ballot for ISO PAS 23617- Healthcare organization management: Pandemic response (respiratory) —Guidelines for medical support of socially vulnerable groups – Comments due 16 October
[Issue 14-99]
Contact: Lee Webster (lswebste@utmb.edu, lwebster@ingenesis.com), Mike Anthony (mike@standardsmichigan.com), Jack Janveja (jjanveja@umich.edu), Richard Robben (rrobben1952@gmail.com), James Harvey (jharvey@umich.edu), Christine Fischer (chrisfis@umich.edu), Dr Veronica Muzquiz Edwards (vedwards@ingenesis.com)
Category: Health, Global
More
ISO Focus Special Issue on Healthcare
ISO/TC 48 Laboratory equipment
ISO/TC 212 Clinical laboratory testing and in vitro diagnostic test systems
ISO/TC 198 Sterilization of health care products
Four years ago Mom made a surprise visit to the ‘Hyacinth Chen School of Nursing’. Was always her dream that young women, especially from poor families, fulfil theirs to become nurses. The students were ecstatic to actually see a lady they only knew as a painting on the wall. pic.twitter.com/LBHHCLVhKy
— Wayne Chen (@wcchen) June 1, 2022
- The American National Standards Institute — the Global Secretariat for ISO — does not provide content management systems for its US Technical Advisory Groups. Because of the nascent committee, inspired by the work of Lee Webster at the University of Texas Medical Branch needed a content management system, we have been managing content on a Google Site facility on a University of Michigan host since 2014.Earlier this spring, the University of Michigan began upgrading its Google Sites facility which requires us to offload existing content onto the new facility before the end of June. That process is happening now. Because of this it is unwise for us to open the content library for this committee publicly. Respecting copyright, confidentiality of ISO and the US Technical Advisory Group we protect most recent content in the link below and invite anyone to click in any day at 15:00 (16:00) UTC. Our office door is open every day at this hour and has been for the better part of ten years.